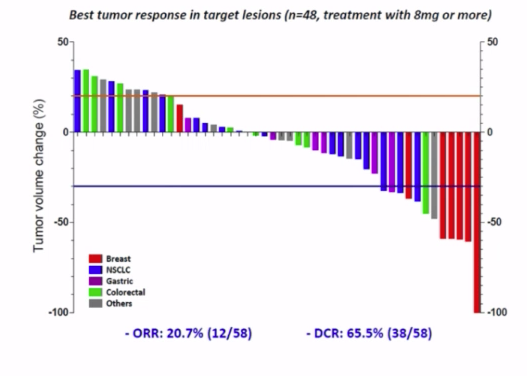
There is currently no FDA approved therapy for patients with NSCLC harboring HER2 exon 20 insertion mutations.Ī study for poziotinib has been initiated to confirm the clinical benefit seen in Cohort 2, as required for an accelerated approval. The agency has set a PDUFA date of November 24, 2022. The NDA is based on the positive results of Cohort 2 in patients with previously treated locally advanced or metastatic non-small cell lung cancer (NSCLC) harboring HER2 exon 20 insertion mutations. The New Drug Application (NDA) for poziotinib is under active review at the FDA with Fast Track designation. 
Poziotinib, a Pan ErbB inhibitor targeting HER2 exon20 mutations The company is working with its partner, Hanmi Pharmaceutical, to support the FDA regulatory review process. Food and Drug Administration (FDA) has accepted Spectrum’s resubmitted Biologics License Application (BLA) for eflapegrastim with a Prescription Drug User Fee Act (PDUFA) date of September 9, 2022. “We are proud of the progress we’ve made toward our core business objectives and we remain dedicated to making a meaningful difference in the lives of cancer patients.” The resubmitted BLA for eflapegrastim was also accepted for review by the FDA,” said Tom Riga, President and Chief Executive Officer of Spectrum Pharmaceuticals. In the first quarter, we initiated a confirmatory study and presented additional positive scientific data for poziotinib. “We anticipate FDA approvals later this year for poziotinib and eflapegrastim. (NasdaqGS: SPPI), a biopharmaceutical company focused on novel and targeted oncology therapies, announced today financial results for the three-month period ended Maand provided a corporate update. HENDERSON, Nev.-( BUSINESS WIRE)-Spectrum Pharmaceuticals, Inc.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
